Feline eosinophilic keratoconjunctivitis is a chronic and progressive infiltrative disease of the cornea and conjunctiva and a crucial differential in cases of raised lesions on the cornea in cats
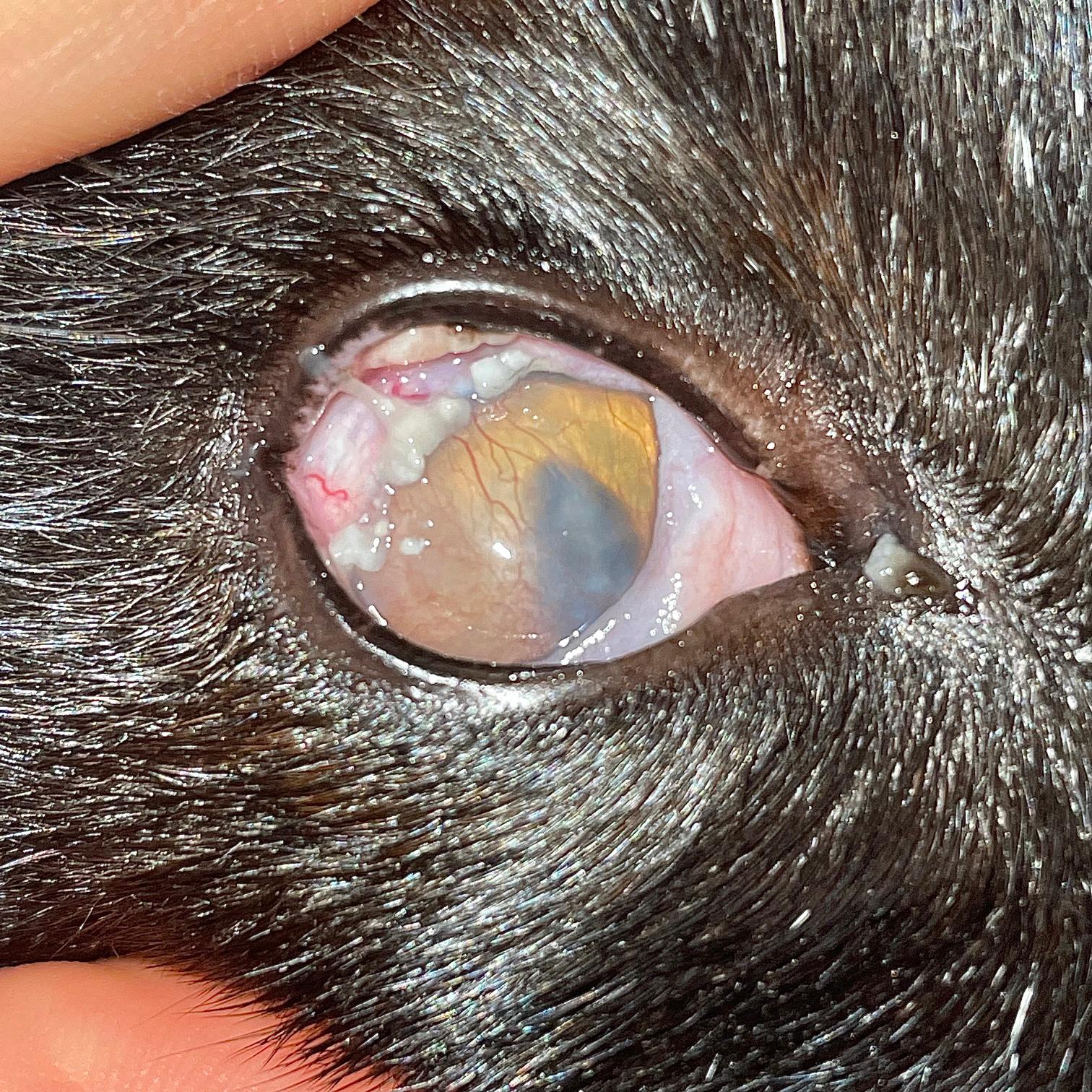
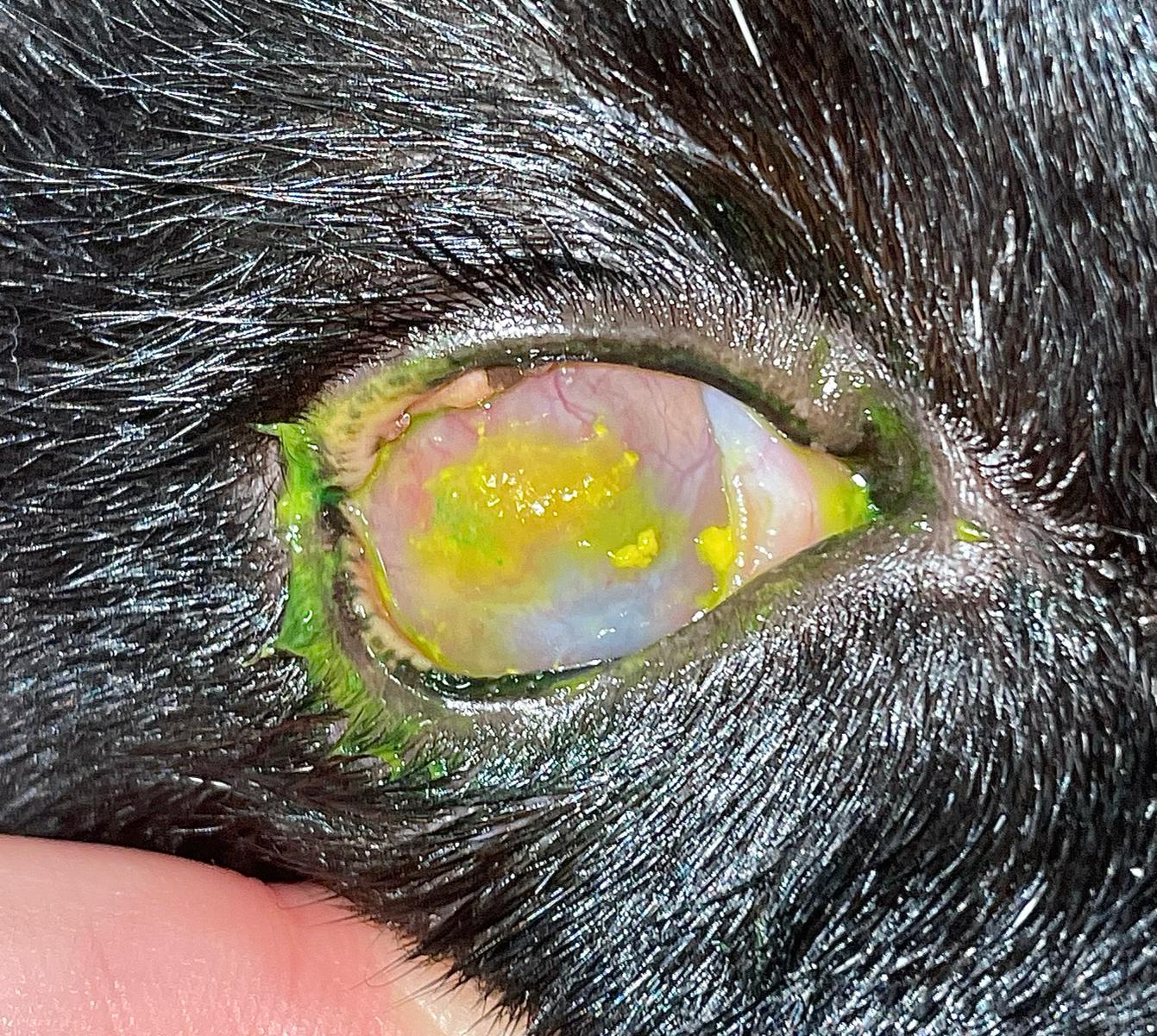
Consider this clinical case: a four-year-old male neutered domestic shorthair cat presents to your clinic with a history of intermittent blepharospasm of the left eye. It has increased seromucoid ocular discharge and what the owner describes as a “pink film” that has grown across its left eye over the past two weeks (Figure 1).

What would you do?
You perform an ophthalmic examination, and there is indeed a strange lesion affecting the cat’s cornea. So you rack your brains on what you might be seeing and come up with the following list:
- Corneal ulcer that is healing with neovascularisation and granulation tissue
- Corneal abscess
- Corneal neoplasia
- Some weird form of keratitis
You need to find out which of these you are seeing. What do you do next? Your options are:
- Fluorescein stain
- Perform corneal cytology
- Start the cat on chloramphenicol (left eye (OS) TID), lubricant (OS BID) and oral meloxicam (SID).
- Any eye case unnerves you – you refer it
You decide to apply fluorescein stain to the eye; it is retained by the pinkish tissue, as well as a small area of adjacent cornea. What do you do now?

Option one: “Ahh,” you say, “this confirms my suspicion of a healing corneal ulcer.” So you proceed with option three and start the cat on the above medications. However, the patient returns in five days, appearing no better, and the pink area seems more extensive.
Option two: “Maybe I should perform cytology anyway?” you consider. Despite knowing that cytology should, ideally, be performed before fluorescein staining, you go ahead with this plan. You see Figure 2 under the microscope. “Hang on,” you exclaim. “I know what this is – it is feline eosinophilic keratoconjunctivitis!”
Now you must decide your next move. What are your thoughts? What must you consider? And what are your next steps?
- I know I need to start some form of immunomodulator, but it’s ulcerated. Will I make it worse?
- I read somewhere that feline herpesvirus has been implicated in corneal ulceration and feline eosinophilic keratoconjunctivitis, so it’s probably likely in this case. If I start steroids, will I precipitate a herpetic flare-up?
- Should I start the cat on Maxidex or Maxitrol OS q12h?
- Should I start the cat on Optimmune (0.2 percent ciclosporin) OS q12h?
- What about chloramphenicol OS q6h?
- What about a topical NSAID (eg ketorolac) OS q12h?
- Or maybe an antiviral treatment such as topical ganciclovir OS QID or oral famciclovir PO BID?
- Should I use a combination of two or more of these treatments?
You decide to prescribe Maxitrol (OS q12h). At a recheck 10 days later, lo and behold, there has been dramatic improvement! There is barely a hint of raised plaques: just a subtle haze of receding vessels and no fluorescein uptake. Basking in the success of your treatment, you advise four more days of Maxitrol (OS q12h) before stopping, and you happily sign the cat off.
Two months later, to your dismay, a now disgruntled owner brings the cat back, complaining that the eye problem seems to have returned with a vengeance. “Did I do something wrong the first time?” you question. You restart Maxitrol – OS q8h this time – but doubts assail you. You decide to ring your friendly neighbourhood ophthalmologist for advice.
Read on to see what they might have to say.
What is feline eosinophilic keratoconjunctivitis?
Feline eosinophilic keratoconjunctivitis (FEK), also called proliferative keratoconjunctivitis, is a unique chronic and progressive infiltrative disease of the cornea and conjunctiva in the cat. It is an important differential in all cases of feline corneal opacification with raised lesions.
Cytology of the affected tissue is usually diagnostic. Immunosuppressive therapy is key to successful treatment; however, there are multiple factors to consider in determining the ultimate treatment strategy. Challenges associated with managing FEK include whether to treat with topical corticosteroids in the face of concurrent corneal ulceration and/or probable feline herpesvirus (FHV-1) involvement, as well as what to do in non-compliant or refractory cases and how to limit or deal with recurrent disease.
Pathogenesis
Although FEK has been reported in veterinary scientific literature since 1979 (Brightman et al., 1979), there are still gaps in our understanding of many aspects of this condition. Its specific aetiology and pathogenesis are uncertain. However, the general consensus is that FEK is an immune-mediated disorder in which there is an exaggerated/aberrant immune response to an (unknown) antigenic stimulus, occurring by way of either a type I or type IV hypersensitivity reaction (Prasse and Winston, 1996).
A type I reaction is IgE mediated, with mast cell and eosinophil degranulation as the cause of tissue damage, whereas type IV reactions are T-lymphocyte mediated, with IL-5 production and tissue damage resulting from recruitment and degranulation of eosinophils.
Is there a link with feline herpesvirus infection?
Veterinary literature reveals that feline herpesvirus (FHV-1) DNA has been recovered using polymerase chain reaction (PCR) from corneal samples in 55 to 76 percent of FEK cases, compared to 5.9 percent of corneas from normal cats (Déan and Meunier, 2013; Nasisse et al., 1998). The presence of FHV-1 DNA was especially prominent in cases where there was precedent or concurrent corneal ulceration, suggesting a strong association between the two conditions.
However, other studies have demonstrated that up to 43 percent of clinically normal-appearing cat corneas also harbour FHV-1 DNA, making its demonstration by PCR difficult to causally relate to specific disease conditions (Stiles et al., 1997; Townsend et al., 2004). It could also be that the corneal inflammation and stress associated with FEK are responsible for the recrudescence of FHV-1 from latency.
Nevertheless, just as stromal keratitis can develop alongside chronic FHV-1 as a delayed-type hypersensitivity response directed at viral antigens sequestered in the corneal stroma (Maggs, 2018; Nasisse et al., 1995), it is plausible that FHV-1 infection has a role in triggering the hypersensitivity response seen in FEK (Labelle and Labelle, 2023), but further studies are needed to test this hypothesis.
Recognising feline eosinophilic keratoconjunctivitis
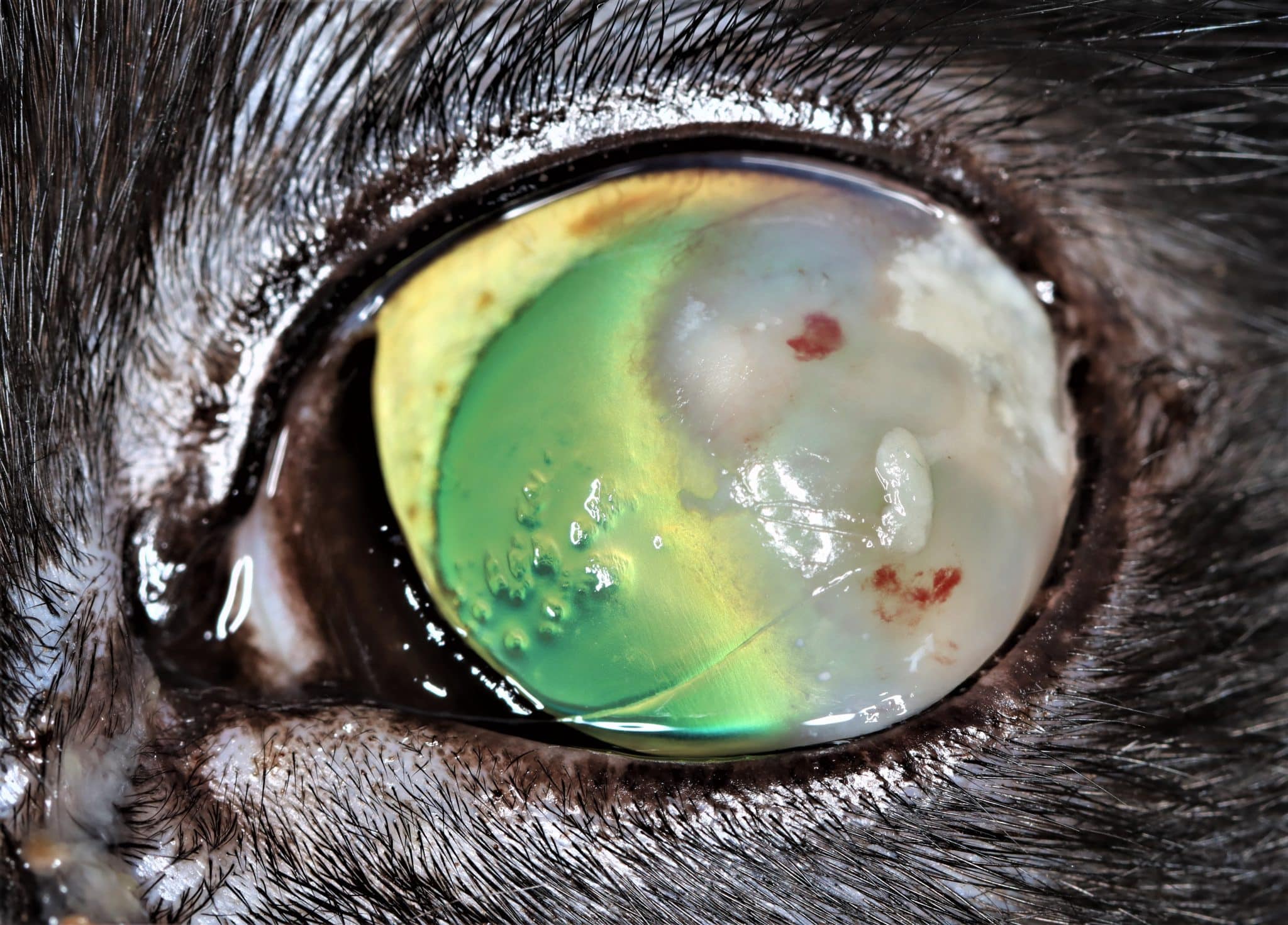
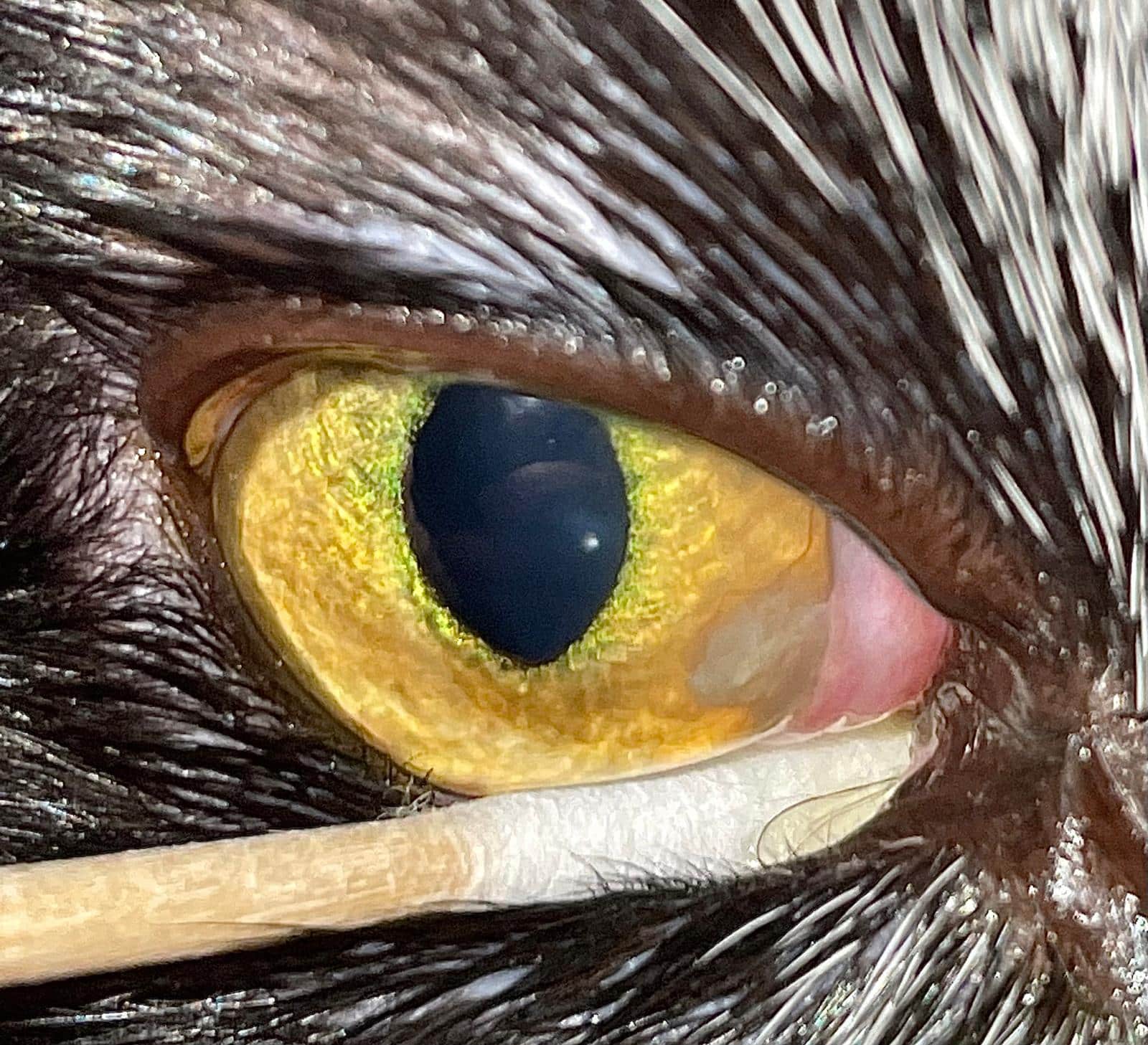
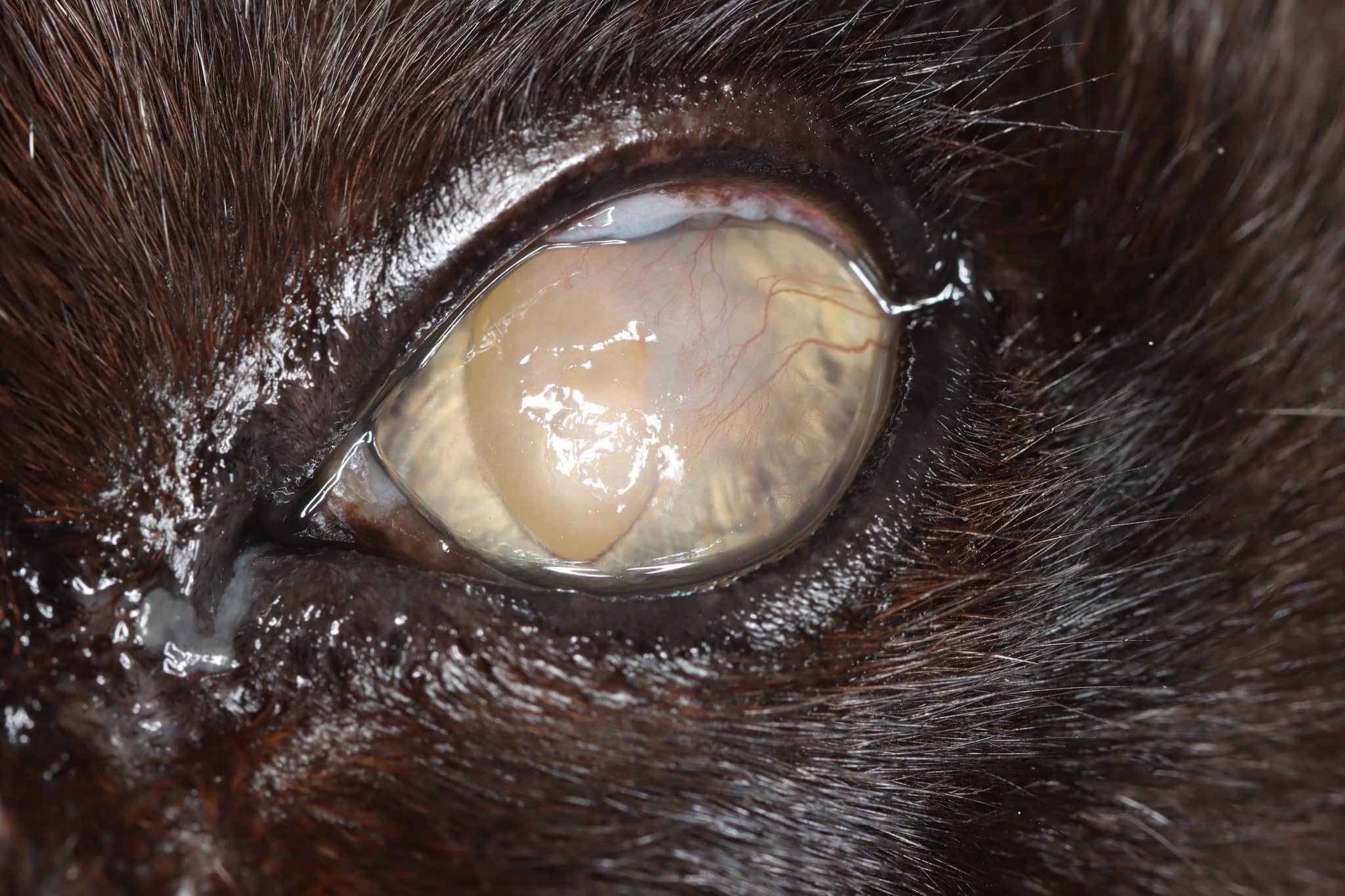
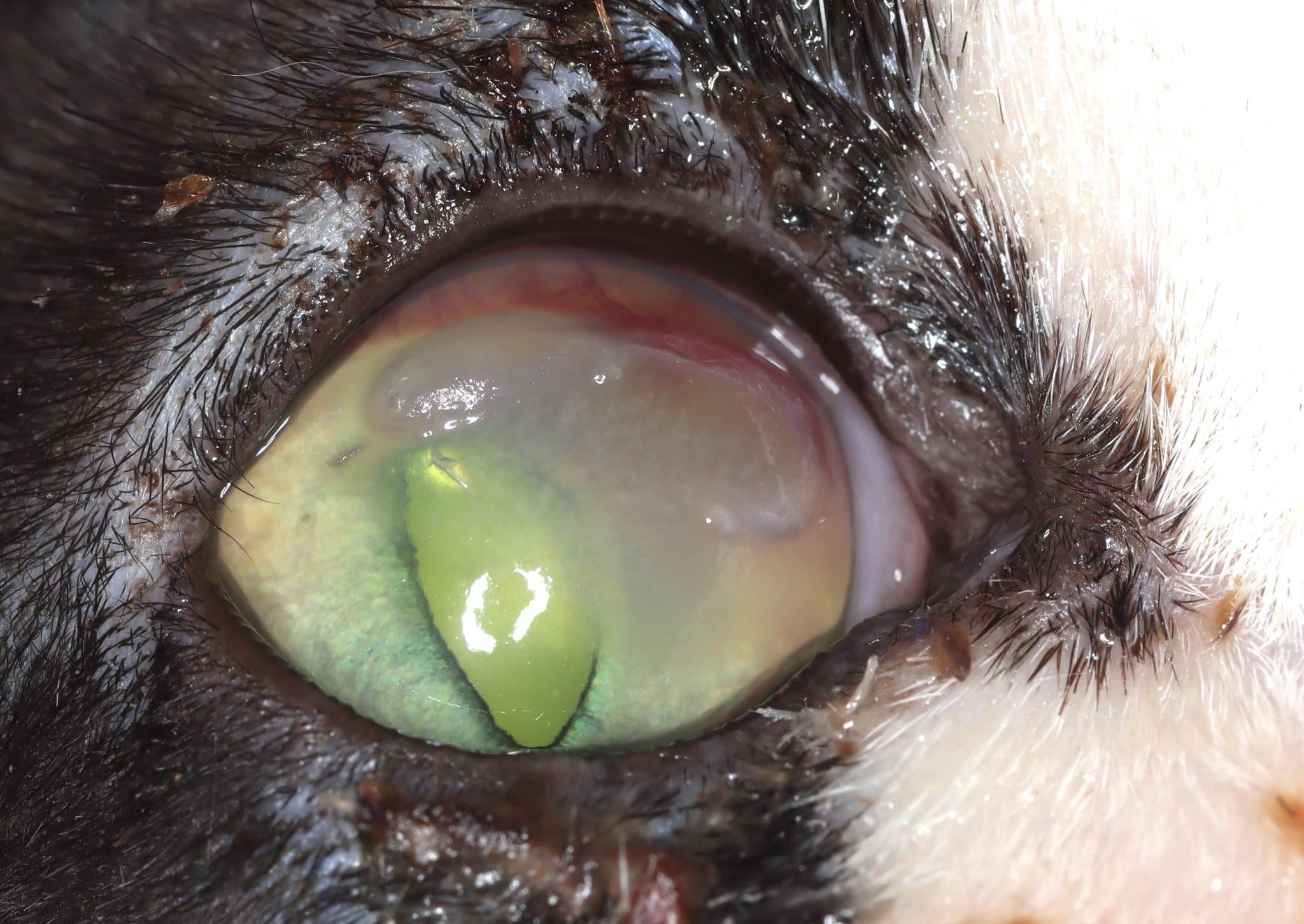
FEK is characterised by progressive corneal neovascularisation that begins perilimbally; cellular corneal infiltration (often with concomitant oedema); and single or multiple focal to coalescing, irregular, raised, white to cream to pink, superficial plaques on the corneal surface (Figure 3). The plaques are a hallmark feature and may be finely granular or flocculent, or resemble either granulation tissue or “cottage cheese” in appearance. Sometimes, FEK can furtively manifest as diffuse corneal infiltrates rather than discrete plaques (Figure 4).
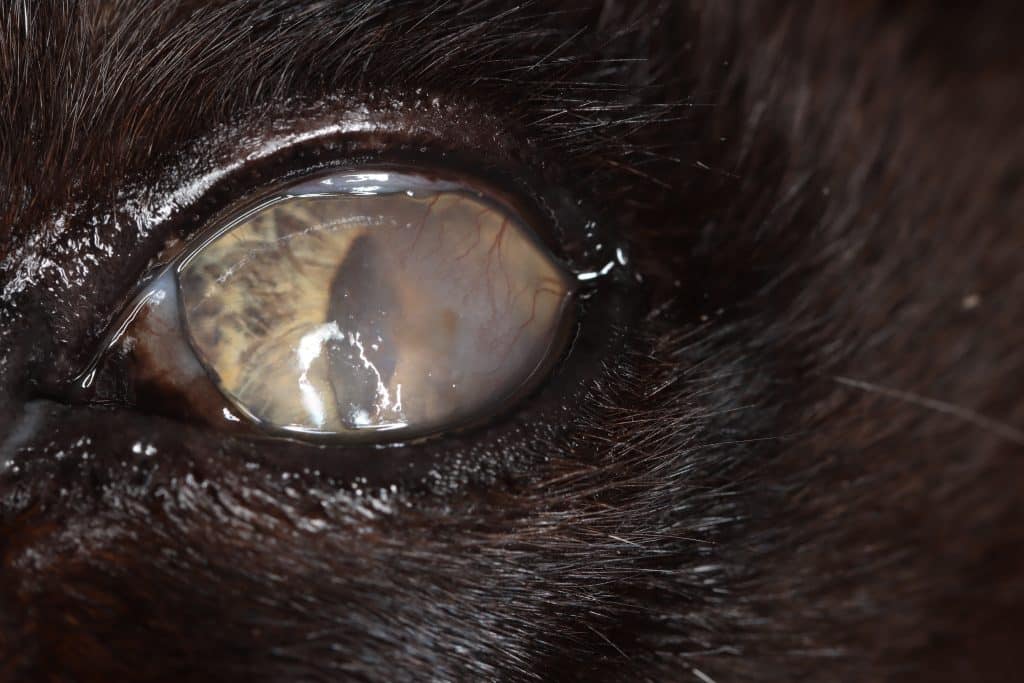
Lesions tend to start at the dorsotemporal (Figures 3, 4, 8 and 9) or ventronasal (Figures 5 and 6) quadrants of the cornea: in 76.8 and 12.5 percent of cases, respectively (Déan and Meunier, 2013; Lucyshyn et al., 2020). However, in advanced cases, the entire cornea may be affected (Figure 7).
Ocular discharge (which may be serous, mucoid or mucopurulent) is also common, and there may also be conjunctival hyperaemia and chemosis, particularly if the adjacent conjunctiva and/or third eyelid become involved (Morgan et al., 1996; Spiess et al., 2009).
Plaques tend to retain fluorescein, and ulceration of the adjacent corneal epithelium may also be present (Figures 8 and 9). In the latter cases, many cats show some degree of ocular discomfort. Otherwise, FEK elicits less blepharospasm than might be expected for the degree of corneal involvement. Additionally, the disease is not generally associated with a clinically detectable reflex uveitis (Labelle and Labelle, 2023). Indeed, some cats may appear asymptomatic, except for a “cloudiness of” or “film over” the eye – a common description from owners.
If the corneal infiltrate/plaques extend over the visual axis, they can compromise vision and quality of life. It can also make visualising the pupil and assessing deeper ocular structures challenging for the clinician. In severely affected cases, using a bright focal light to assess for a dazzle reflex is useful as an indicator of retinal function.
FEK is most commonly unilateral (66 to 75.6 percent of cases) but may also be or become bilateral (Déan and Meunier, 2013; Paulsen et al., 1987; Spiess et al., 2009).
Signalment
Young to middle-aged neutered male cats were reported to be most commonly affected in some studies (Lucyshyn et al., 2020; Morgan et al., 1996; Spiess et al., 2009). However, this condition is not bound by signalment.

Corneal ulceration and feline eosinophilic keratoconjunctivitis
In cases of FEK, epithelial loss over an eosinophilic plaque and the surrounding cornea is common. Corneal ulceration has been noted in about 30 percent of eyes at the time of FEK diagnosis(Déan and Meunier, 2013; Stiles and Coster, 2016; Spiess et al., 2009). One study also found that cats with either corneal ulceration or a history of it had a higher percentage of positive FHV-1 PCR samples (67 percent) than cats with no corneal ulcer at any time (22 percent) (Déan and Meunier, 2013).
Although this finding cannot be interpreted specifically as FHV-1 leading to the ulceration, it is certainly interesting. In humans with atopic keratoconjunctivitis, the presence of extracellular toxic eosinophil protein deposits was documented and found to interfere with corneal epithelial cell viability and migration (Messmer et al., 2002). It may be that a similar process, in which eosinophils in the plaques release epitheliotoxic substances that lead to ulceration, occurs in cats with FEK.
An ulcer caused by degranulated eosinophil toxins would improve with a topical corticosteroid addressing the inflammatory infiltrate, while an ulcer caused by active FHV-1 might deteriorate if treated similarly. Ultimately, whether corneal ulceration in FEK occurs secondary to FHV-1, eosinophilic inflammation or other cytokine-induced cytolysis is difficult to ascertain and may differ from case to case.
Nevertheless, having this background understanding may enable clinicians to rationalise their treatment strategy. This includes whether it is relatively safe to use a topical corticosteroid or whether antiviral treatment might be called for, depending on the degree of clinical suspicion of FHV-1 involvement.
Differential diagnoses
The case scenario above alludes to the reality that, despite its relatively characteristic clinical appearance, not all FEK cases are textbook-level pathognomonic and so can sometimes be mistaken for other conditions.
Common differentials are discussed in Table 1 (Balicka et al., 2021; Morgan et al., 1996; Paulsen et al., 1987).
| Condition | Appearance | Comments |
|---|---|---|
| Granulation tissue secondary to chronic ulcerative keratitis (ie a healing corneal injury) |
|
FEK is usually suspected when there is a history of non-response to antibiotic treatment |
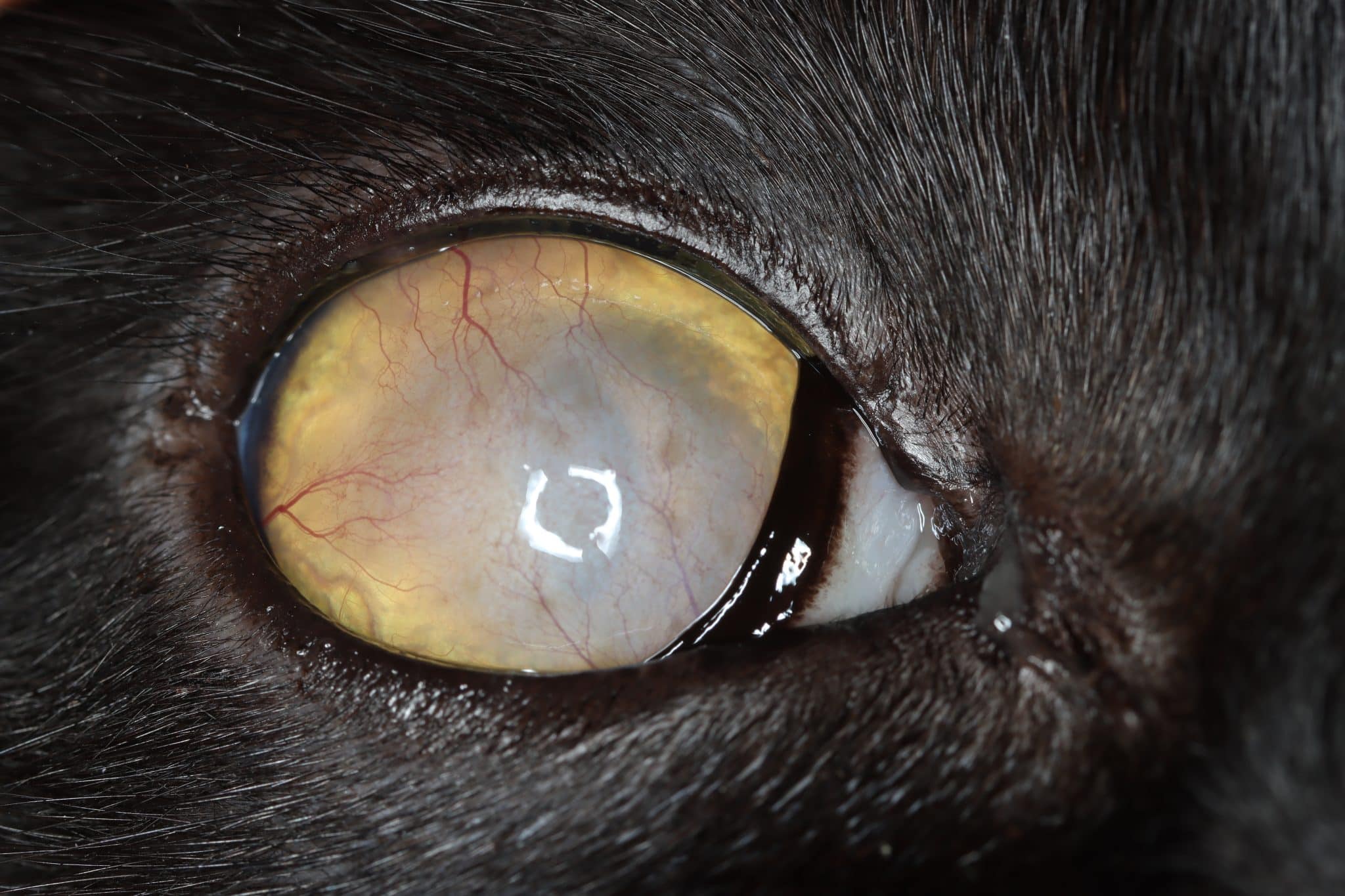
| FHV-1 keratitis (stromal keratitis) |


|
Treatment usually involves an antiviral agent and a topical broad-spectrum antibiotic if ulceration is
present
Corticosteroids are generally contraindicated during active infection, however judicious use may be called for as stromal keratitis is an immunopathogenic form of chronic FHV-1 disease |
| Symblepharon | For examples see Figure 28.36 in Gelatt et al., 2021 (p. 1713) or Figures 3A or 4B from Shiraishi et al., 2023 |
Adhesions of the palpebral, bulbar and/or nictitans conjunctiva to itself or to the cornea can cause corneal
opacification
Tends to affect kittens/juvenile cats, as its development is frequently associated with primary FHV-1 infection |
| Corneal stromal abscess |


|
Typically fluorescein-negative stromal lesions that are creamy-white to yellow in colour due to neutrophilic infiltration, with notable oedema, vascularisation and discomfort (Gelatt et al., 2021) |
| Foreign body granuloma | n/a | Where a granulomatous inflammatory reaction develops in response to an exogenous foreign body or an endogenous substance |
| Acid-fast granuloma (ie mycobacterial keratitis) | For examples see Figure 138 in Ketring and Glaze, 2012 (p. 69) or Figure 6 from Stavinohova et al., 2019 |
Develops insidiously as a nodular vascularised mass, usually with associated corneal oedema
Observation of acid-fast bacilli on histopathology of affected tissue is diagnostic (Maggs, 2018) |
| Fungal keratitis (keratomycosis) | For examples see Figure 28.41 from Gelatt et al., 2021 (p. 1723) or Figure 136 in Ketring and Glaze, 2012 (p. 68) |
Infections by filamentous fungi are usually initiated by trauma. Those due to yeast-like fungi usually develop secondary to existing ocular problems
There is often unusually severe blepharospasm. Raised, opaque surface plaque(s) may be seen with ulceration, secondary corneal vascularisation and anterior uveitis Diagnosed by cytological evidence of fungal hyphae in corneal scrapings or fungal culture and PCR (Gelatt et al., 2021) |
| Corneal degeneration/deposits |

|
Can occur secondary to corneal injury or chronic inflammation. The deposits may be calcium, lipid or drug plaques |
| Corneal neoplasia (eg squamous cell carcinoma or lymphoma) | For examples see Figure 1 from Scurrell et al., 2013 or Figure 164 in Ketring and Glaze, 2012 (p. 68) |
Other species, including dogs, horses and cows, are more often affected by corneal squamous cell carcinoma, but there are only three reports in the scientific literature of corneal squamous cell carcinoma in the cat and none of feline corneal lymphoma (Delgado, 2020; Perlmann et al., 2010; Scurrell et al., 2013)
In contrast to FEK, corneal neoplasia is rare and should not be a top differential as this could lead to erroneous recommendation of enucleation, which would never be indicated with FEK |
What’s next?
Stay tuned for part two of this series, wherein we discuss diagnostic tests and treatment strategies for feline eosinophilic keratoconjunctivitis.
Acknowledgements
The author would like to thank the ophthalmology team at North Downs Specialist Referrals, in particular: Dr Emily Jeanes for helping proofread and edit the article; Dr Adam Margetts, Dr Vim Kumaratunga, Dr Richard Everson, Dr Emily Jeanes and Dr Andra Enache, for the photographs; and all the above ophthalmologists for illuminating discussions on FEK.
Case Advice or Arranging a Referral
If you are a veterinary professional and would like to discuss a case with one of our team, or require pre-referral advice about a patient, please call 01883 741449. Alternatively, to refer a case, please use the online referral form
About The Discipline
Ophthalmology

Need case advice or have any questions?
If you have any questions or would like advice on a case please call our dedicated vet line on 01883 741449 and ask to speak to one of our Ophthalmology team.
Advice is freely available, even if the case cannot be referred.
Ophthalmology Team
Our Ophthalmology Team offer a caring, multi-disciplinary approach to all medical and surgical conditions.